Inside the Liposomal Moment: Practical Specs, Real Manufacturing, and What Buyers Actually Ask
I’ve spent enough time with formulators and brand owners to know this isn’t just hype anymore. The shift to Liposomal delivery is real—driven by consumers who want “it works faster” without mega-doses, and by manufacturers who need stability, cleaner labels, and fewer customer complaints about GI upset. From vitamins to nootropics, the playbook’s changing.

What’s trending (and why it matters)
Three quick trends: (1) smaller particles (≈80–180 nm D50) via high-pressure microfluidization; (2) non-GMO sunflower phosphatidylcholine replacing soy; (3) tougher stability testing—brands now ask for ICH-like data and real-time plus accelerated. To be honest, it’s overdue.
Core technicals—what buyers check first
| Parameter | Typical spec (≈) | Method / Standard |
|---|---|---|
| Particle size (D50) | 100–150 nm | DLS per ISO 22412 |
| PDI | DLS | |
| Encapsulation efficiency | ≥ 80% | Dialysis + HPLC |
| Zeta potential | -20 to -40 mV | Electrophoretic light scattering |
| Microbial / Heavy metals | Meets limits | USP /, USP ICP-MS |
| Shelf life | 24 months at 2–8°C (real-world may vary) | ICH Q1A(R2) |
How a solid Liposomal line is actually made
- Materials: non-GMO phosphatidylcholine (sunflower), cholesterol (stability), purified water, actives (vitamin C, glutathione, curcumin, NAD+, iron, etc.).
- Methods: thin-film hydration → high-pressure microfluidization (or ethanol injection) → size reduction → sterile filtration if applicable.
- Testing: DLS for size/PDI; HPLC potency; encapsulation efficiency; pH/osmolality; microbial; heavy metals; stability (real-time + 40°C/75% RH accelerated).
- Certifications: cGMP, ISO 22000/9001, HACCP; optional Halal/Kosher.
- Service life: typically 18–24 months refrigerated; RTD beverages may differ based on pH and preservatives.
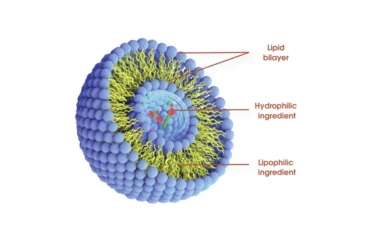
Where Liposomal shines
Use cases: nutraceutical capsules and liquids, functional drinks (clear or lightly turbid), dermal serums (phospholipid skin affinity), pediatric/geriatric formats, and even veterinary supplements. Many customers say tolerance improves versus high-dose conventional powders—less “acid bite” for vitamin C, for instance.
Vendor comparison (what buyers quietly benchmark)
| Criteria | Finutra (Zhengding, Hebei) | Typical OEM | Commodity Supplier |
|---|---|---|---|
| cGMP / ISO 22000 | Yes | Varies | Rare |
| Microfluidizer control | Tight (D50 targets) | Moderate | Limited |
| Customization (actives, flavor, pH) | High | Medium | Low |
| Stability data (ICH-like) | Available | Sometimes | No |
Customization and packaging
Options include vegan phospholipids, sugar-free flavor systems, clear beverage-grade dispersions, or viscous gels for sachets. Packaging: HDPE bottles, glass, or unit-dose sachets; nitrogen headspace for sensitive actives. Origin: Building 23B1, No.2 Yuanboyuan St., Zhengding Area of China (Hebei) Pilot Free Trade Zone.
Mini case studies
- EU vitamin C liquid: moved to Liposomal, cut serving size by ~40% with equal plasma exposure (internal comparative data; n=16; real-world results vary).
- APAC beauty brand: Liposomal retinol serum reduced irritation reports and maintained assay within 90–110% after 6 months at 25°C.
- US RTD nootropic: switched to Liposomal curcumin; improved dispersion (D50 ~120 nm) and 9‑month clarity stability at pH 3.4.
Why teams pick it
Better bioavailability potential, protection from gastric/oxidative stress, and—surprisingly—cleaner flavor profiles when formulated right. I guess the short version is: fewer compromises.
Authoritative references
- FDA. Guidance for Industry: Liposome Drug Products (2018).
- Torchilin VP. Recent advances with liposomes as pharmaceutical carriers. Nat Rev Drug Discov. 2005.
- Lasic DD. Liposomes: From Physics to Applications. Elsevier, 1993.
- EFSA Scientific Committee. Guidance on risk assessment of nanoscience applications in food/feed (2021).
- ICH Q1A(R2). Stability Testing of New Drug Substances and Products.
Post time:Oct - 24 - 2025

























